Sofie at Work: How One CDMO Succeeded Across 3 Sites
For this article of Sofie at Work, see how one CDMO leverages Sofie across multiple sites.

TransferAI Team
Sofie at Work is our series where biopharma professionals share real workflows of Sofie in action. This article follows a pharmaceutical organization's multi-site pilot program, where teams across quality, regulatory, IT, and operations discovered that the right AI platform doesn't just save time—it becomes indispensable.
When working across multiple sites and offices, precision matters even more as teams juggle method verifications, deviation investigations, SOP consolidation, and regulatory research. Even with precise, professional teams, the stakes are high and doubt can linger when teams are chasing updates, consolidating spreadsheets, and losing hours to work that doesn't move anything forward.
Sofie changes that. And this multi-site team proved it by piloting Sofie across three sites, where they’ve experienced early adoption, measurable cost savings, cross-departmental impact, and expanded capacity for high-value work. Sofie produced significant tangible wins in less than 60 days across departments proving that Sofie had a direct and positive impact on budgets and operations.
The Results: Proof Across Departments
Let’s walk through the wins this multi-site pilot observed and the time savings across each use case.
Streamlining Multi-Day Method Verification
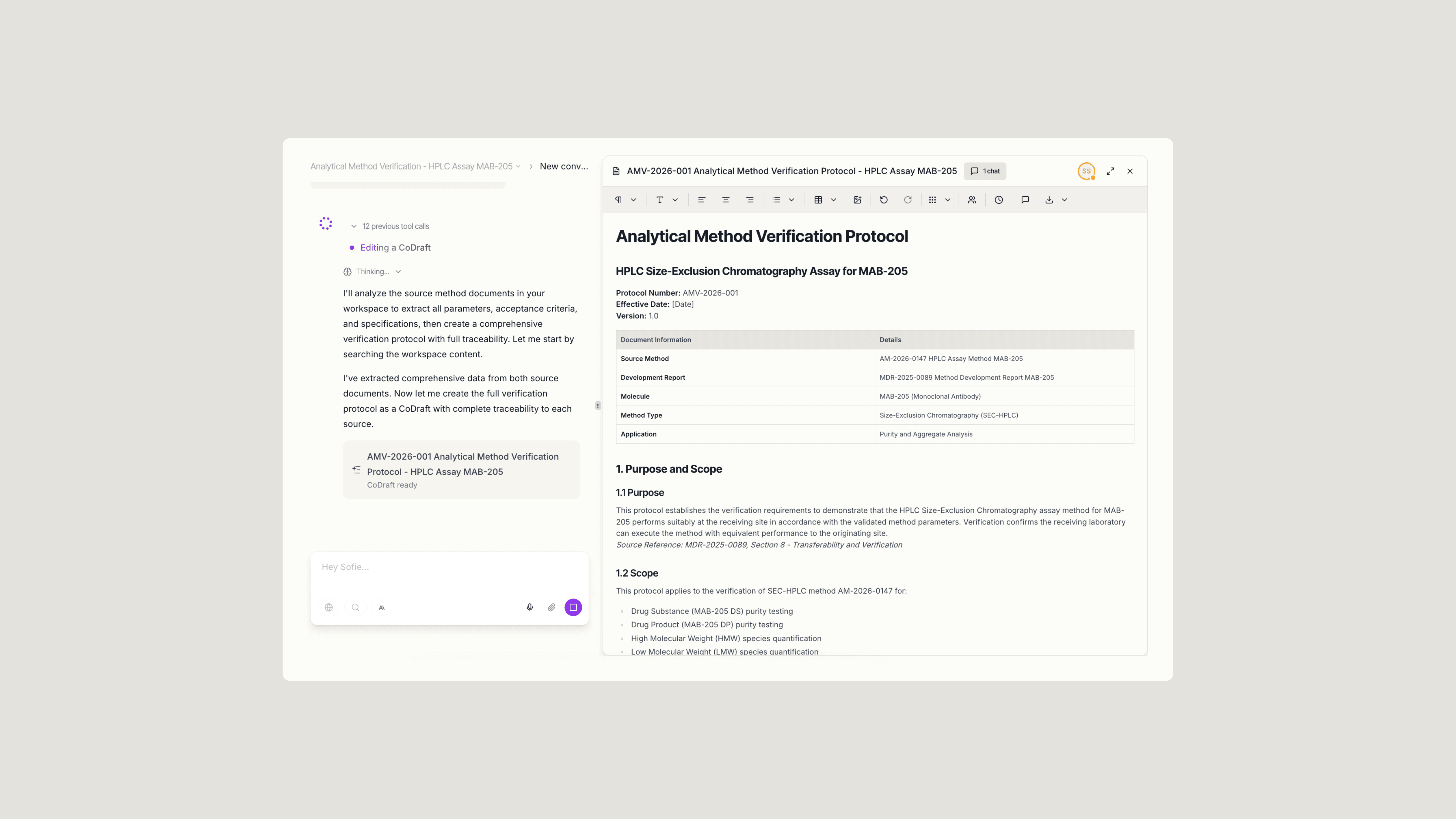
Upload your source method and prompt Sofie to generate a complete verification protocol. In this pilot, one team uploaded their analytical method documentation and asked for a full protocol covering parameters, acceptance criteria, and test specifications.
Sofie analyzed the source method and returned a structured draft with each section sourced back to the original document. What previously took 2-3 days of manual drafting dropped to 10 minutes. This way, you're able to focus on reviewing and refining the protocol rather than drafting from scratch.
Cutting IT Admin from Hours to Minutes
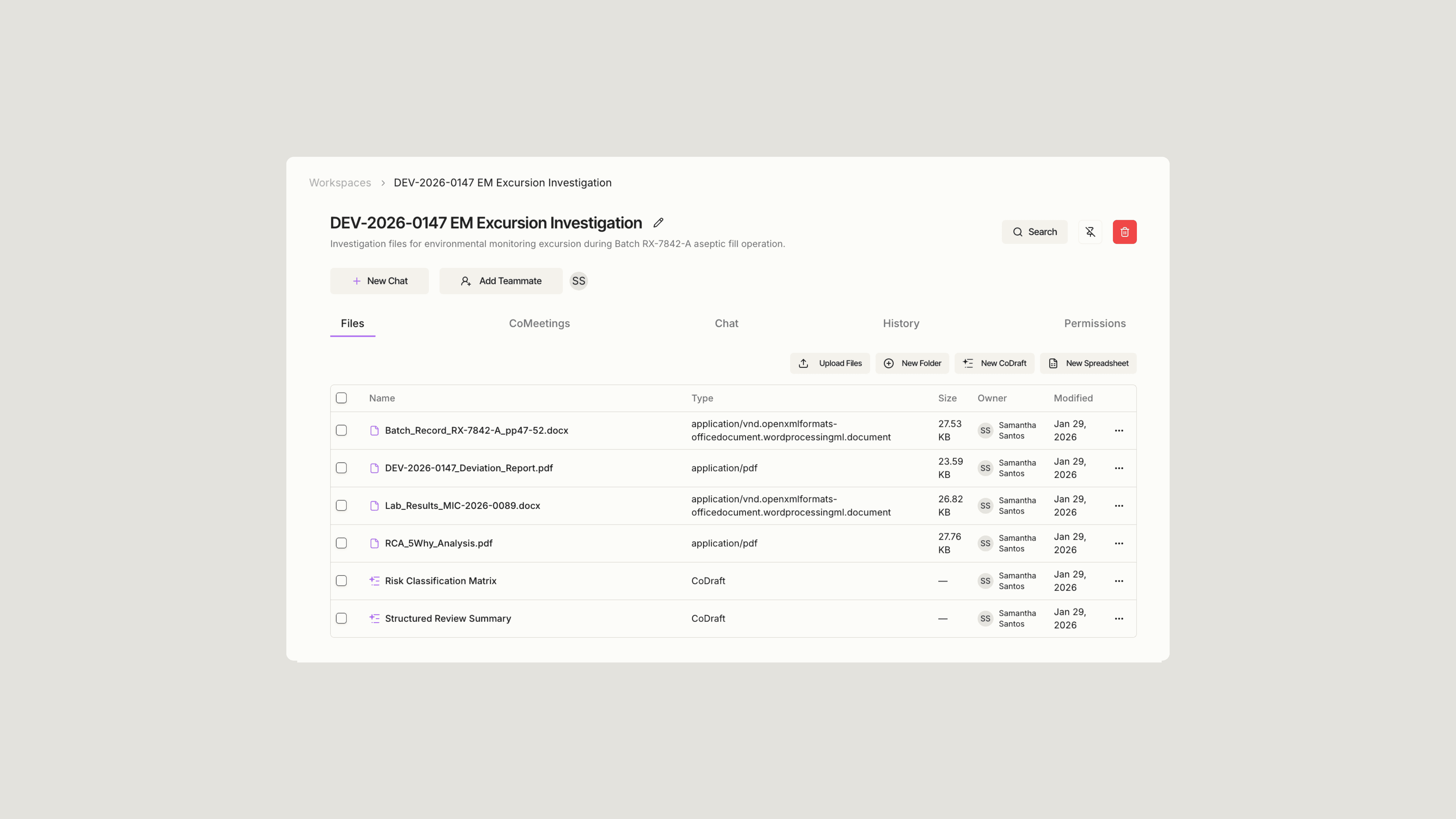
Sofie compressed a labor-intensive weekly review—6+ hours of manual cross-referencing—down to 15 minutes of structured, organized output. Instead of assembling data from multiple sources and hunting for discrepancies, the team now receives a consolidated view ready for action. That's real capacity recovered: hours every week that IT staff can redirect toward system improvements, process optimization, and strategic projects rather than repetitive data reconciliation.
Accelerating Investigation Reviews
Upload your deviation investigation documentation and prompt Sofie to structure your review. Quality teams in the pilot uploaded their investigation files and asked for problem statements, timelines, scope assessments, and risk classifications.
Sofie analyzed the documentation and returned organized feedback each element traced back to the source material. Reviews that took 45-60 minutes dropped to 10 minutes. Teams were able to refocus on decision-making and resolution rather than document assembly.
Regulatory Research in Half the Time
Upload your regulatory questions and relevant guidance documents, then prompt Sofie to research and summarize. Teams in the pilot asked specific questions about FDA compliance requirements and let Sofie dig through the guidance.
Sofie surfaced relevant passages and organized findings with citations back to source documents. Research tasks that required 4-6 hours were completed in 2 hours. The team was then able to focus on applying the guidance for their active FDA review.
Faster Protocol Review
Upload your protocol documents and prompt Sofie to review for corrections. In the pilot, 60% of documents required only simple corrections like formatting issues, inconsistencies, missing sections.
Sofie flagged issues and suggested corrections, each linked to the specific section. The 2-3 hour redlining process dropped to a fraction of the time. Sofie did the legwork so the team could focus on verifying and approving changes for streamlined review.
Project Management Without the Busywork
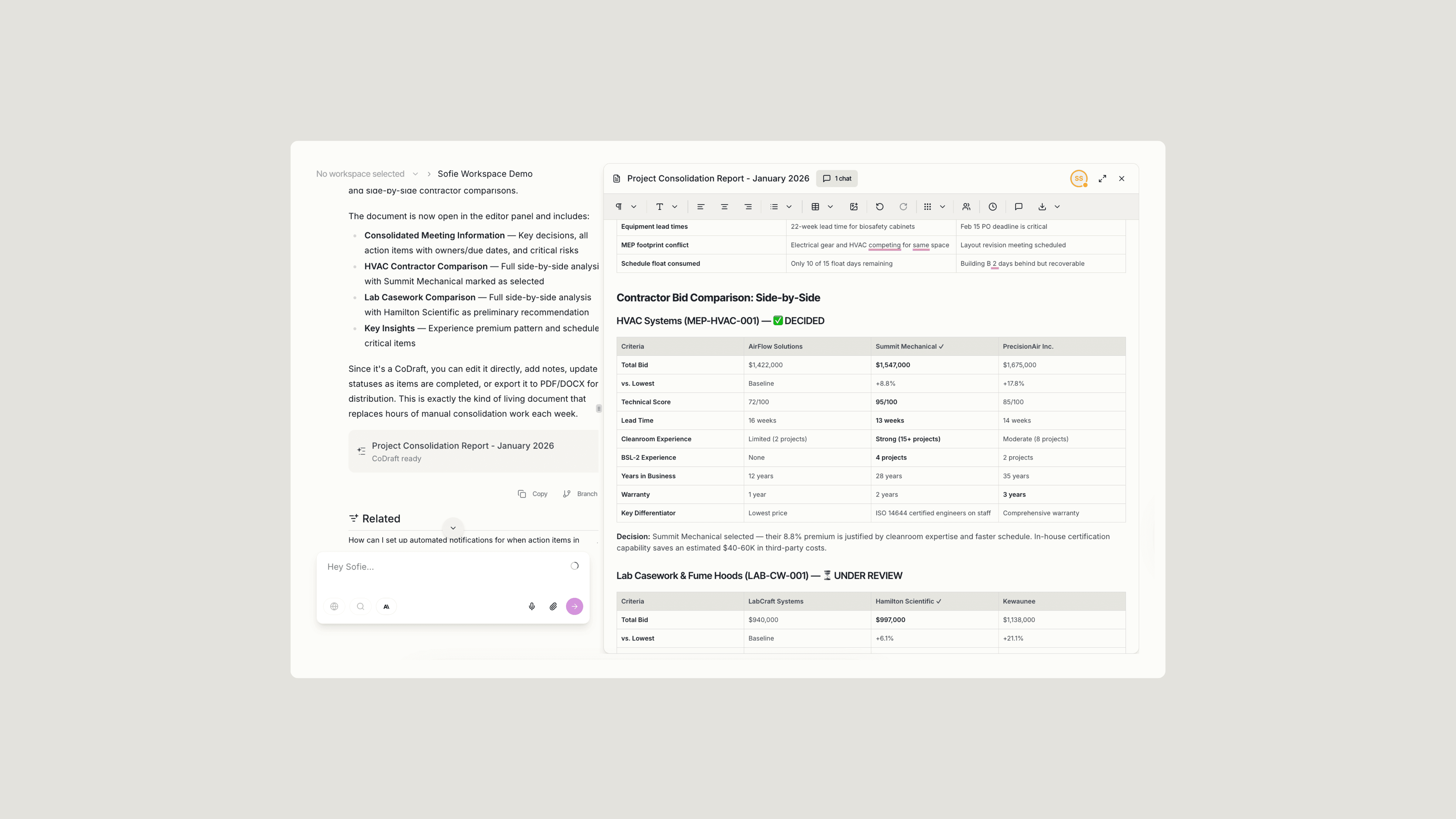
Upload your project status documents, meeting notes, or contractor bids and prompt Sofie to organize and analyze. Teams in the pilot used Sofie to track statuses across workstreams, consolidate meeting information, and compare contractor bids side-by-side.
What previously required hours of manual consolidation saved approximately 3 hours per week. The team was able to stay actively focused on driving decisions and moving the project forward.
Beyond Efficiency
The strongest AI implementations aren't measured by time saved alone—they're measured by adoption that sticks. Teams discovered applications no one initially scoped:
Risk assessments — Structured risk evaluations for cleaning methodologies and process changes
Handwritten batch record extraction — Pulling data from handwritten entries into usable formats
French-English translation — Bidirectional translation of technical documents for multi-site coordination
RFP analysis — Comparing customer RFPs against templates, identifying gaps, drafting proposal content
Certificate generation — Creating COAs, COTs, and other compliance certificates
Sofie was purpose-built for biopharma: embedded industry knowledge, memory that persists across sessions, direct vendor access, and a commitment to stand with the organization during FDA audits. Generic tools couldn't match that.
The goal isn't replacement. It's giving teams back the hours lost to routine work so they can focus on the decisions that require human expertise. The result is that Sofie works the way your teams actually work—compliance and productivity built in from the start, with both operations and quality aligned and moving faster.
If you want a fresh set of high-impact ideas tailored to your use of Sofie, request a 1:1.